当前位置:
首页 »
资料 »
药物合成路线 » Loxoprofen sodium, CS-600G, LX-A, CS-600, Loxonin,80382-23-6
Loxoprofen sodium, CS-600G, LX-A, CS-600, Loxonin,80382-23-6

更新日期:2018-11-23 浏览次数:
443
摘 要:Loxoprofen sodium, CS-600G, LX-A, CS-600, Loxonin,80382-23-6, 68767-14-6 (free acid),C15-H17-O3.Na,(?-2-[4-(2-Oxocyclopentylmethyl)phenyl]propionic acid sodium salt
- 【药物名称】Loxoprofen sodium, CS-600G, LX-A, CS-600, Loxonin
- 【化学名】(?-2-[4-(2-Oxocyclopentylmethyl)phenyl]propionic acid sodium salt
- 【CAS登记号】80382-23-6, 68767-14-6 (free acid)
- 【结构式】
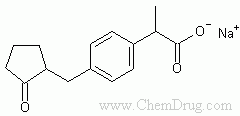
- 【分子式】C15-H17-O3.Na
- 【分子量】268.2863
- 【原研厂家】Sankyo (Originator), Lead Chemical (Codevelopment), Toko Pharmaceutical (Codevelopment)
- 【作用类别】Antiarthritic Drugs, TREATMENT OF MUSCULOSKELETAL & CONNECTIVE TISSUE DISEASES, Non-Steroidal Antiinflammatory Drugs
- 【研发状态】Launched-1986
- 【合成情况】
- 〖来源〗Drugs Fut
- 〖合成路线〗
- 〖标题〗Loxoprofen
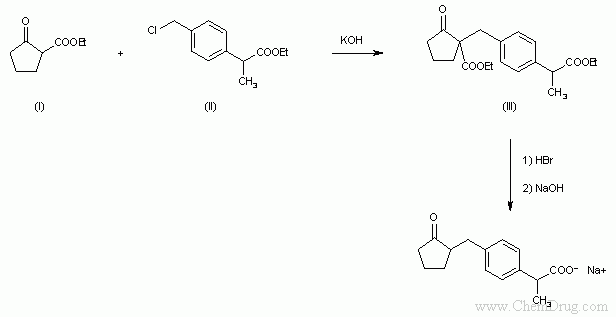
- 〖合成方法〗The reaction of ethyl 2-oxocyclopentanecarboxylate (I) with ethyl 2-(4-chloromethylphenyl)propionate (II) by means of KOH in hot DMF gives ethyl 2-[4-(1-ethoxycarbonyl-2-oxocyclopentan-1-ylmethyl)phenyl]propionate (III), which is then hydrolyzed and decarboxylated by treatment with 47% HBr in refluxing dioxane.
- 〖作者〗Castaer, J.; Serradell, M.N.
- 〖参考〗Castaer, J.; Serradell, M.N.; Loxoprofen. Drugs Fut 1984, 9, 1, 22
- 〖出处〗Drugs Fut1984,9,(1):22
- 〖备注〗Synthesis of 127127:
The reaction of ethyl 2-oxocyclopentanecarboxylate (I) with ethyl 2-(4-chloromethylphenyl)propionate (II) by means of KOH in hot DMF gives ethyl 2-[4-(1-ethoxycarbonyl-2-oxocyclopentan-1-ylmethyl)phenyl]propionate (III), which is then hydrolyzed and decarboxylated by treatment with 47% HBr in refluxing dioxane (1). (Scheme 12712701a)
Description
Free acid, m.p. 108.5-11.0? b.p.(0.3) 190-5?(1).
Manufacturer
Sankyo (Japan).
Reference
1. Terada, A., Wachi, K. and Misaka, E (Sankyo Co., Ltd.); US 4.161.538.
- 〖来源〗DE 2814556; ES 468578; FR 2395256; GB 1580113; US 4161538
- 〖合成路线〗

- 〖标题〗Substituted phenylacetic acid derivatives and process for the preparation thereof
- 〖合成方法〗The reaction of ethyl 2-oxocyclopentanecarboxylate (I) with ethyl 2-(4-chloromethylphenyl)propionate (II) by means of KOH in hot DMF gives ethyl 2-[4-(1-ethoxycarbonyl-2-oxocyclopentan-1-ylmethyl)phenyl]propionate (III), which is then hydrolyzed and decarboxylated by treatment with 47% HBr in refluxing dioxane.
- 〖作者〗Misaka, E.; Terada, A.; Wachi, K. (Sankyo Co., Ltd.)
- 〖参考〗Misaka, E.; Terada, A.; Wachi, K. (Sankyo Co., Ltd.); Substituted phenylacetic acid derivatives and process for the preparation thereof. DE 2814556; ES 468578; FR 2395256; GB 1580113; US 4161538
- 〖出处〗DE 2814556; ES 468578; FR 2395256; GB 1580113; US 4161538,,():
- 〖备注〗Synthesis of 127127:
The reaction of ethyl 2-oxocyclopentanecarboxylate (I) with ethyl 2-(4-chloromethylphenyl)propionate (II) by means of KOH in hot DMF gives ethyl 2-[4-(1-ethoxycarbonyl-2-oxocyclopentan-1-ylmethyl)phenyl]propionate (III), which is then hydrolyzed and decarboxylated by treatment with 47% HBr in refluxing dioxane. (Scheme 12712701a)
Description
Free acid, m.p. 108.5-11.0? b.p.(0.3) 190-5?
本文导航:
-
(1)
Loxoprofen sodium, CS-600G, LX-A, CS-600, Loxonin,80382-23-6







