当前位置:
首页 »
资料 »
医药中间体库 » ,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]a
,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]a
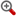

更新日期:2018-11-23 浏览次数:
207
摘 要:,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]amino)-5-[(6-methyl-3-pyridinyl)methyl]-4(3H)-pyrimidinone
- 【化学名】2-([2-[(2-Furylmethyl)sulfanyl]ethyl]amino)-5-[(6-methyl-3-pyridinyl)methyl]-4(3H)-pyrimidinone
- 【CAS登记号】
- 【结构式】
![,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]a--药物合成数据库](/file/upload/SYNTHESIS/STR/100/10003.gif)
- 【分子式】C18H20N4O2S
- 【分子量】356.4497
- 【来源】
- 【合成情况】
- 〖目标产物〗Lupitidine hydrochloride, SK&F-93479
- 〖合成路线〗

- 〖合成方法〗A new synthesis of lupitidine has been described:
The condensation of 5-(2-methylpyridin-5-yl)-2-(nitroamino)pyrimidin-4(3H)-one (I) with 2-(2-furylmethylthio)ethylamine (II) in refluxing pyridine gives 2-[2-(2-furylmethylthio)ethylamino]-5-(2-methylpyridin-5-ylmethyl)pyrimidin-4(3H)-one (III), which is then submitted to a conventional Mannich condensation with formaldehyde and dimethylamine in acetic acid.
- 〖参考〗Parsons, M.E.; Rawlings, D.A.; Ife, R.J.; Brown, T.H.; Blakemore, R.C.; Blurton, P.; Armitage, M.A.; Durant, G.J.; Slingsby, B.P.; Ganellin, C.R.; Isocytosine H2-receptor histamine antagonists. III. The synthesis and biological activity of lupitidine (SK&-Methylpyridin-5-yl)-2-(nitroamin?肩?b`数据库????F:\vb\chemdr
本文导航:
-
(1)
,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]a
![,C18H20N4O2S,356.4497,2-([2-[(2-Furylmethyl)sulfanyl]ethyl]a--药物合成数据库](/file/upload/SYNTHESIS/STR/100/10003.gif)





