当前位置:
首页 »
资料 »
医药中间体库 » ,C5H5N3,107.1157,2-(1H-Imidazol-1-yl)acetonitrile
,C5H5N3,107.1157,2-(1H-Imidazol-1-yl)acetonitrile

更新日期:2018-11-23 浏览次数:
468
摘 要:,C5H5N3,107.1157,2-(1H-Imidazol-1-yl)acetonitrile
- 【化学名】2-(1H-Imidazol-1-yl)acetonitrile
- 【CAS登记号】
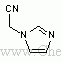
- 【结构式】
- 【分子式】C5H5N3
- 【分子量】107.1157
- 【来源】
- 【合成情况】
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗
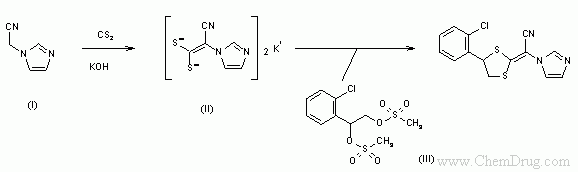
- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Castaer, J.; Prous, J.; Lanoconazole. Drugs Fut 1993, 18, 3, 222
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗

- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Seo, A.; Kanno, H.; Hasegawa, N.; Miyagi, Y.; Nishimura, A.; Konaka, S.; Ohmi, T.; Munechika, Y.; Uchida, M.; Ikeda, K. (Nihon Nohyaku Co., Ltd.); Antimycotic agent and fungicidal agent. US 4738976
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗

- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Seo, A.; Sugano, H.; Hasegawa, C.; Ikeda, K.; Munechica, Y.; Konoe, T.; Konaka, M. (Nihon Nohyaku Co., Ltd.); Antifungal agent. JP 1987093227
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗

- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Seo, A.; Sugano, H.; Hasegawa, C.; Ikeda, K.; Nishimura, A.; Miyashiro, Y. (Nihon Nohyaku Co., Ltd.); Non-medicinal bactericidal agents and method for their preparation. JP 1987093204
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗

- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Seo, A.; Sugano, H.; Hasegawa, C.; Miyashiro, Y.; Nishimura, A.; Ikeda, K. (Nihon Nohyaku Co., Ltd.); Ketene S,S-acetals. JP 1985218387
- 〖目标产物〗Latoconazole, Lanoconazole, TJN-318, NND-318, Astat
- 〖合成路线〗

- 〖合成方法〗The reaction of 2-(1-imidazolyl)acetonitrile (I) with CS2 and KOH in DMF gives the dithiolate (II), which is then cyclized with 1-(2-chlorophenyl)-1,2-di(methanesulfonyloxy)ethane (III). A column chromatography over silicagel allows the separation of the (E)- and (Z)-isomers.
- 〖参考〗Seo, A.; Kanno, H.; Hasegawa, N.; Miyagi, Y.; Nishimura, A.; Konaka, S.; Ohmi, T.; Munechika, Y.; Uchida, M.; Ikeda, K. (Nihon Nohyaku Co., Ltd.); A novel ketene S,S-acetal deriv., a process for manufacturing thereof and a method for curing mycosis by admarget="_blank" title="Clic
- 〖目标产物〗Luliconazole, PR-2699, NND-502
- 〖合成路线〗
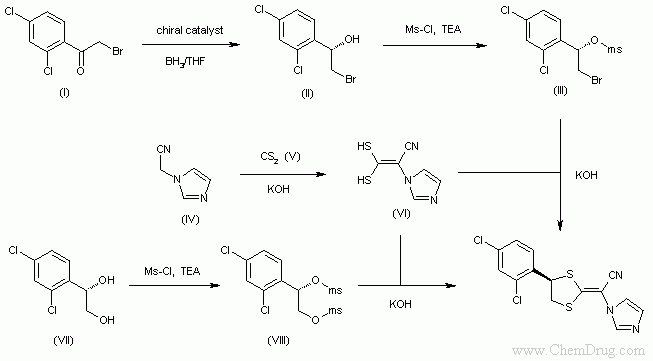
- 〖合成方法〗This compound has been obtained by two similar ways: The enantiocontrolled reduction of 2,4-dichlorophenacyl bromide (I) with BH3/THF and a chiral oxazaborole catalyst gives 1(S)-(2,4-dichlorophenyl)-2-bromoethanol (II), which is treated with Ms-Cl and TEA in dichloromethane to yield the corresponding mesylate (III). Finally, this compound is cyclized with 2-(1-imidazolyl)-3,3-disulfanylacrylonitrile (VI) by means of KOH in DMSO.
Alternatively, 1(S)-(2,4-dichlorophenyl)ethane-1,2-diol (VII) is treated with Ms-Cl and TEA in dichloromethane to give the corresponding dimesylate (VIII), which is condensed with the acrylonitrile (VI) with KOH as before.
The intermediate acrylonitrile (VI) has been obtained by condensation of 2-(1-imidazolyl)acetonitrile (IV) with CS2 (V) by means of KOH in DMSO.
- 〖参考〗Kodama, H.; Niwano, Y.; Kanai, K.; Yoshida, M. (Nihon Nohyaku Co., Ltd.); Antifungal agent, cpd. therefor, process for producing the same. JP 1997100279; JP 1998158168; US 5900488; WO 9702821
本文导航:
-
(1)
,C5H5N3,107.1157,2-(1H-Imidazol-1-yl)acetonitrile












