当前位置:
首页 »
资料 »
医药中间体库 » ,C9H12O6,216.1924,(5R)-5-[(4S)-2,2-dimethyl-1,3-dioxolan-4-y
,C9H12O6,216.1924,(5R)-5-[(4S)-2,2-dimethyl-1,3-dioxolan-4-y
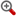

更新日期:2018-11-23 浏览次数:
249
摘 要:,C9H12O6,216.1924,(5R)-5-[(4S)-2,2-dimethyl-1,3-dioxolan-4-yl]-3,4-dihydroxy-2(5H)-furanone
- 【化学名】(5R)-5-[(4S)-2,2-dimethyl-1,3-dioxolan-4-yl]-3,4-dihydroxy-2(5H)-furanone
- 【CAS登记号】
- 【结构式】
- 【分子式】C9H12O6
- 【分子量】216.1924
- 【来源】
- 【合成情况】
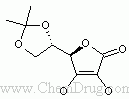
- 〖目标产物〗Campestanol ascorbyl phosphate
- 〖合成路线〗
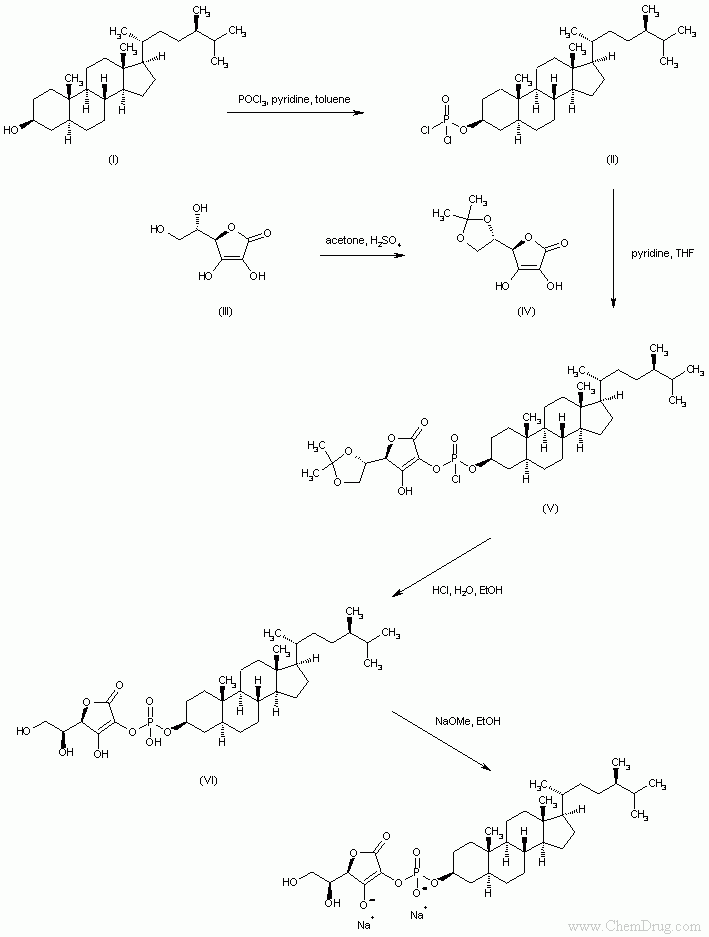
- 〖合成方法〗Reaction of campestanol (I) with phosphorus oxychloride and pyridine gives the dichlorophosphoryl compound (II). Ascorbic acid (III) is protected as the 5,6-isopropylidene ketal (IV) upon treatment with acetone and fuming sulfuric acid. Condensation of (IV) with acid chloride (II) provides the phosphoryl monochloride (V), which is hydrolyzed under acidic conditions to yield the phosphate diester (VI). Finally, compound (VI) is converted to the target disodium salt by using an ethanolic solution of NaOMe. (1,2)
- 〖参考〗Chen, H.; Milanova, R.K.; Kutney, J.P.; Hou, D.; Ding, Y. (Forbes Medi-Tech Inc.); Conjugates of phytosterol or phytostanol with ascorbic acid and use thereof in treating or preventing cardiovascular disease. JP 2003522124; WO 0100653
- 〖目标产物〗Campestanol ascorbyl phosphate
- 〖合成路线〗

- 〖合成方法〗Reaction of campestanol (I) with phosphorus oxychloride and pyridine gives the dichlorophosphoryl compound (II). Ascorbic acid (III) is protected as the 5,6-isopropylidene ketal (IV) upon treatment with acetone and fuming sulfuric acid. Condensation of (IV) with acid chloride (II) provides the phosphoryl monochloride (V), which is hydrolyzed under acidic conditions to yield the phosphate diester (VI). Finally, compound (VI) is converted to the target disodium salt by using an ethanolic solution of NaOMe. (1,2)
- 〖参考〗Novak, E.; Milanova, R.K.; Kutney, J.P.; Pritchard, H.P.; Lukic, T. (Forbes Medi-Tech Inc.); Novel structures comprising phytosterol and/or phytostanol and ascorbic acid and use thereof as weight regulating agents. WO 0234241
- 〖目标产物〗Sitostanol ascorbyl phosphate
- 〖合成路线〗
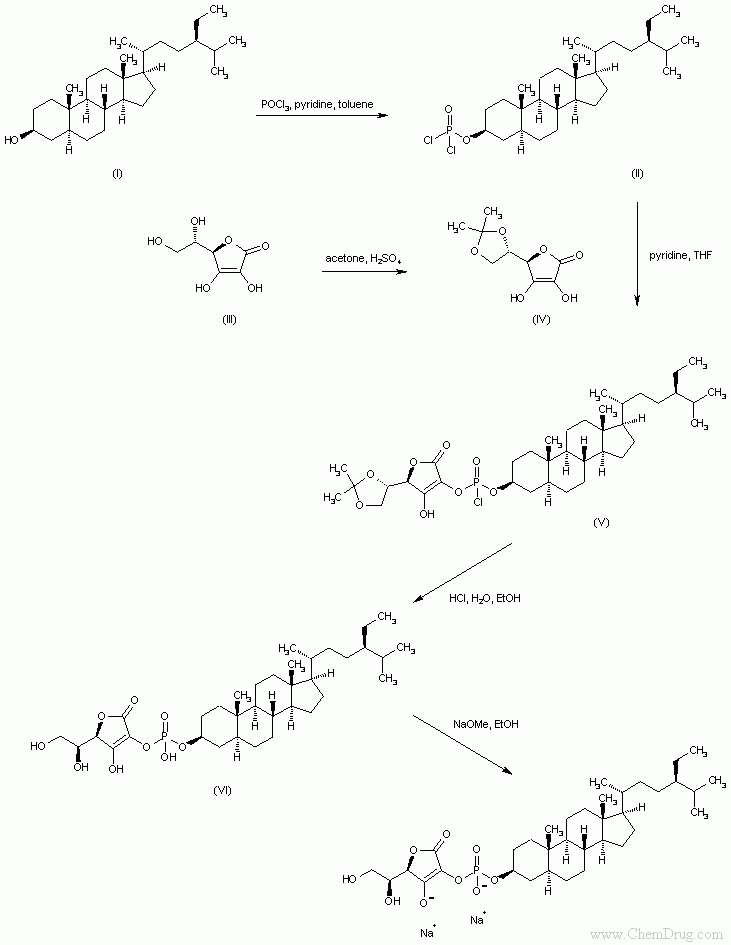
- 〖合成方法〗Reaction of sitostanol (I) with phosphorus oxychloride and pyridine affords the dichlorophosphoryl compound (II). Ascorbic acid (III) is protected as the 5,6-isopropylidene ketal (IV) upon treatment with acetone and fuming sulfuric acid. Condensation of (IV) with acid chloride (II) provides the phosphoryl monochloride (V), which is hydrolyzed under acidic conditions to yield the phosphate diester (VI). Finally, compound (VI) is converted to the target disodium salt by using an ethanolic solution of NaOMe. (1,2)
- 〖参考〗Chen, H.; Milanova, R.K.; Kutney, J.P.; Hou, D.; Ding, Y. (Forbes Medi-Tech Inc.); Conjugates of phytosterol or phytostanol with ascorbic acid and use thereof in treating or preventing cardiovascular disease. JP 2003522124; WO 0100653
- 〖目标产物〗Sitostanol ascorbyl phosphate
- 〖合成路线〗

- 〖合成方法〗Reaction of sitostanol (I) with phosphorus oxychloride and pyridine affords the dichlorophosphoryl compound (II). Ascorbic acid (III) is protected as the 5,6-isopropylidene ketal (IV) upon treatment with acetone and fuming sulfuric acid. Condensation of (IV) with acid chloride (II) provides the phosphoryl monochloride (V), which is hydrolyzed under acidic conditions to yield the phosphate diester (VI). Finally, compound (VI) is converted to the target disodium salt by using an ethanolic solution of NaOMe. (1,2)
- 〖参考〗Novak, E.; Milanova, R.K.; Kutney, J.P.; Pritchard, H.P.; Lukic, T. (Forbes Medi-Tech Inc.); Novel structures comprising phytosterol and/or phytostanol and ascorbic acid and use thereof as weight regulating agents. WO 0234241
本文导航:
-
(1)
,C9H12O6,216.1924,(5R)-5-[(4S)-2,2-dimethyl-1,3-dioxolan-4-y









