当前位置:
首页 »
资料 »
医药中间体库 » ,C28H35F3N6O2,544.6254,1-[(4,6-dimethyl-5-pyrimidinyl)carbon
,C28H35F3N6O2,544.6254,1-[(4,6-dimethyl-5-pyrimidinyl)carbon
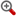

更新日期:2018-11-23 浏览次数:
166
摘 要:,C28H35F3N6O2,544.6254,1-[(4,6-dimethyl-5-pyrimidinyl)carbonyl]-4-((3S)-4-{(1R)-2-methoxy-1-[4-(trifluoromethyl)phenyl]ethyl}-3-methylpiperazinyl)-4-piperidinecarbonitrile
- 【化学名】1-[(4,6-dimethyl-5-pyrimidinyl)carbonyl]-4-((3S)-4-{(1R)-2-methoxy-1-[4-(trifluoromethyl)phenyl]ethyl}-3-methylpiperazinyl)-4-piperidinecarbonitrile
- 【CAS登记号】
- 【结构式】
- 【分子式】C28H35F3N6O2
- 【分子量】544.6254
- 【来源】
- 【合成情况】
- 〖目标产物〗Sch-417690, SCH-D
- 〖合成路线〗
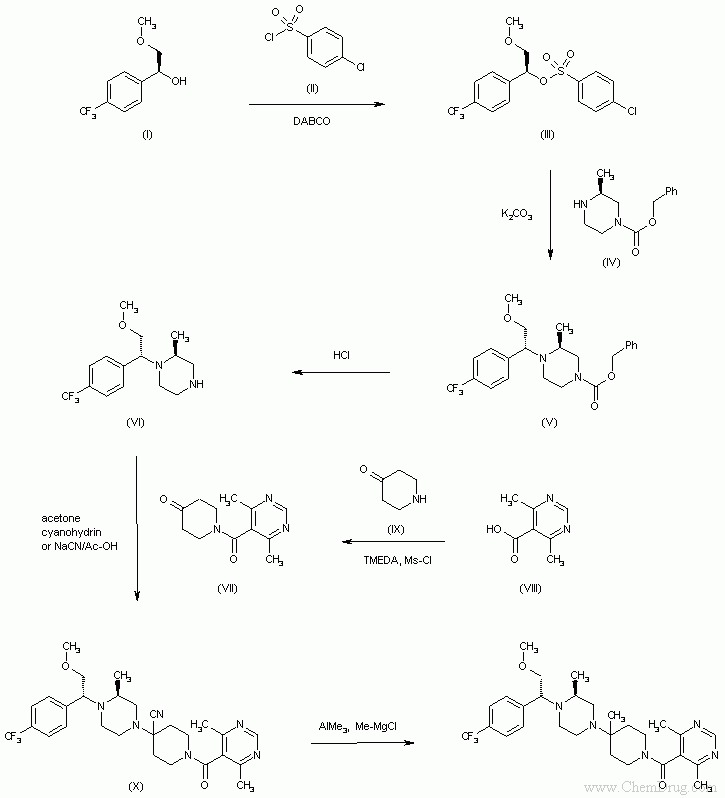
- 〖合成方法〗The reaction of the chiral benzyl alcohol (I) with 4-chlorobenzenesulfonyl chloride (II) and DABCO in toluene gives the sulfonate (III), which is condensed with the monoprotected piperazine (IV) by means of K2CO3 in hot toluene/acetonitrile to yield the adduct (V). The deprotection of (V) by means of hot aqueous HCl affords the piperazine derivative (VI), which is condensed with the acyl piperidine (VII) --obtained by condensation of 4,6-dimethylpyrimidine-5-carboxylic acid (VIII) with 4-piperidone (IX) --by means of acetone cyanohydrin or NaCN/AcOH to provide the cyano adduct (X). Finally, this compound is treated with AlMe3 and Me-MgCl in THF/toluene to yield the target compound.
- 〖参考〗Gala, D.; Leong, W.; Jones, A.D.; Shi, X.; Chen, M.; D'sa, B.A.; Zhu, M.; Xiao, T.; Tang, S.; Goodman, A.J.; Nielsen, C.M.; Lee, G.M.; Gamboa, J.A. (Schering Corp.); Synthesis of piperidine and piperazine cpds. as CCR5 antagonists. WO 0384950
本文导航:
-
(1)
,C28H35F3N6O2,544.6254,1-[(4,6-dimethyl-5-pyrimidinyl)carbon






